SCALE- HOW IS SCALE DETRIMENTAL TO BOILERS?
In this article, we will answer how is scale detrimental to boilers. But first, let’s take a moment to understand scale and the differences between different types.
What Is Water Hardness?
Water hardness (also referred to as “Hardness of water”) is a measurement in parts per millions (ppm) of calcium and magnesium in the water. Water containing calcium and magnesium typically accounts for the majority of boiler scale. In boiler water a level of 0 ppm of calcium and magnesium hardness is desirable but very difficult to achieve. Hard water is treated several different ways in an effort to minimize scale formation in boilers. It is important to remove hardness before entering the boiler because once it is formed it is difficult to remove and causes substantial efficiency losses and potentially damage. There are other natural salts and minerals such as silica that can also cause scale.
How does temporary hardness of water differ from permanent hardness?
Temporary hardness is a type of hardness that can be reduced by heating the water. Temporary hardness primarily made up of calcium carbonate and calcium bicarbonate. These compounds are naturally occurring in water as a result of the action of acidic rainwater on natural minerals. Temporary hardness is also known as alkaline hardness because the hardness dissolves to form an alkaline solution. Permanent hardness is a type of hardness that can be reduced only by the use of chemicals or distillation. Permanent hardness consists of non-carbonate salts.
How is scale detrimental to boilers?
Scale will form around the heating surfaces and less heat will pass through the boiler tubes into the water. Even light accumulation of scale in a boiler causes boiler efficiency to drop. Scale results in higher fuel costs due to increased fuel consumption in order to satisfy the same steam demand. Heavy accumulation of scale can severely impede heat transfer and water can no longer adequately remove the heat from the boiler tubes. The tubes can overheat, soften and burst.
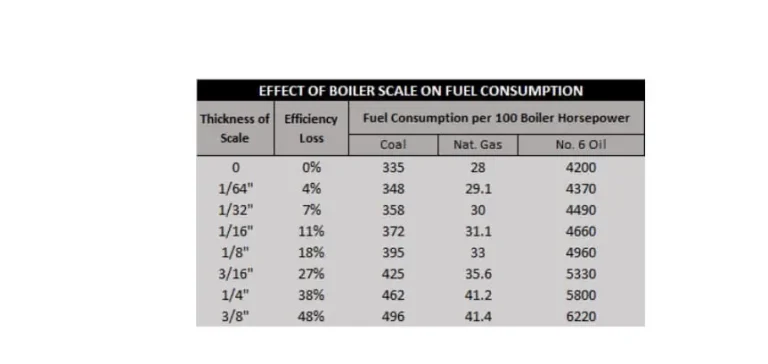
What causes hardness in water to form scale on steam boiler surfaces?
Calcium and magnesium hardness crystallizes and becomes scale in two ways. One way is through Inverse Solubility, which is the tendency of certain impurities in the water to crystallize and precipitate as the temperature of boiler water increases. The hottest water is located against the heating surface of the boiler which causes scale to form in these areas first. The second way is when steam is formed and only pure water is boiled off.
Scale forming impurities are left behind in the boiler while pure water steam is created. When large amounts of water are turned into steam, there are more impurities left behind. Large impurities in the boiler water can become too concentrated forming scale and other problems.
How do you treat for scale?
There are several methods for treating scale the two most common types of treatment are
* Pretreatment- water treatment processes that occur before the water enters the boiler.
* After-Treatment – water treatment processes that occur during or after steam generation.
Complete Water Solutions focuses on mechanical Pretreatment solutions. There are many mechanical means to help reduce scale-forming impurities prior to feedwater tank. We will talk about a few of the most common that are used today:
* Water Softener (Zeolite Water Softener) – Water softeners is an ion-exchange process that uses resin beads and brine solution to soften water. When the softener is in operation the hard water (calcium & magnesium impurities) flow down (in most applications) through the resin. The calcium and magnesium stick to the resin media and release a sodium ion. Once all the resin beads are spent or no longer able to exchange calcium & magnesium for sodium-ion the softener must go through the regeneration process. During this process sodium at concentrated levels remove the calcium and magnesium impurities off of the resin bead. This allows the softener to once again produce soft water.


Industrial Softener System Fleck 2900 Softener System
* Dealkalizer – Dealkalizer is an ion-exchange system that works in a manner similar to sodium zeolite water softeners but exchanges anions rather than cations. Anions such as carbonates and bicarbonates are removed by ion exchange and replaced with chloride.
* Deionization– Demineralizers are an ion-exchange process generally used in high-pressure boiler applications. Deionization typically has either two separate columns of resin (cation & anion) or a mix bed column which will have both resins in the tank. The cation resin is regenerated with sulfuric acid instead of salt. This resin is used to remove calcium, magnesium, iron and other cation impurities from the makeup water. The Anion resin is the dealkalization process that removes carbonates, bicarbonates, sulfates, and other anions from the makeup water. Anion resin is regenerated with caustic soda and removes carbonates, bicarbonates, sulfates, and other anion impurities.
* Reverse Osmosis – RO is a process in which removes undesirable materials from water by using pressure to force the water through a semi-permeable membrane. This process is called “reverse” osmosis because the pressure forces the water to flow in the reverse direction to the flow direction in the process of natural osmosis. RO removes ionized salts, colloids, and organic molecules down to a molecular weight of 100.
Over the last decade Reverse Osmosis has become the pretreatment of choice to help in the reduction of scale in boiler feed water make up. If you don’t have a RO System currently and would like to see if your system could benefit give Complete Water Solutions a call for a free evaluation. Return on investment may be even quicker with our leasing options.
Complete Water Solutions provides all the above equipment and more to help in the reduction of hardness for your boiler system.
Already have equipment? Need Parts or Service?
We Service All Major Manufacturers. Contact us today!

With over 30 years of hands-on experience in the water treatment industry, Nathan Olszak is a trusted water treatment expert. He specializes in designing, engineering, and servicing water treatment systems. As the owner of Complete Water Solutions, Osmonics, and Membrane Cleaning Pro, Nathan has built a reputation for delivering customized water treatment solutions that enhance efficiency, reliability, and water quality across various industries, including the medical, commercial, and manufacturing sectors.
Nathan’s journey in water treatment began as a service technician. He gained in-depth knowledge by working with all major brands of equipment, including Veolia, Suez, Bruner, Culligan, Pentair, Aquamatic, Osmonics, GE Water, Marlo, Lakeside, Fleck, Autotrol, US Filter, ION Pure, Siemens, Evoqua, and many others. This hands-on expertise, combined with his technical certifications, has made him a leader in water system engineering and water management.
Credentials & Expertise
- Certifications: David H. Paul Reverse Osmosis Certification, PLC Programming
- License: Power Plant Operating Engineer 3rd Class
- Specialties:
- Design & engineering of water treatment systems and advanced water systems
- Custom water filtration systems and solutions
- Installation, automation, and repair of water treatment systems
- Expertise in Reverse Osmosis (RO), Deionization, Water Softening, Carbon Filtration, Iron Filtration, UV Treatment, Ultrafiltration (UF), Process Filtration, CEDI/EDI, and more
- Specialized knowledge in Boiler Feed Water, Tower Makeup Water, and drinking water production for industries such as bottling, pharmaceuticals, and food processing
- Project management and reclaiming of water system waste for other uses
- Evaluation of raw water sources and potential contamination risks
- Industrial water treatment strategies to prevent corrosion and optimize system performance
- Boiler water treatment to enhance energy efficiency and system longevity
Nathan’s commitment to excellence extends beyond equipment sales and service. He works closely with clients to develop tailored water treatment services that optimize performance and meet their operations’ unique demands. His expertise covers methods that mitigate water impurities, ensure fresh water accessibility, and maintain regulatory compliance. It also includes the design of high-efficiency reverse osmosis systems for industrial use.
Nathan focuses on the latest technology in water conditioning, water softeners, filters, and pumps to improve maintenance and efficiency. His experience in laboratory testing and biofilm control ensures the highest industry standards in water treatment processes and water management.
Additionally, his deep understanding of infrastructure and water data analysis provides long-term, cost-effective solutions that promote health and safety. His expertise ensures that businesses receive top-tier, quality water treatment solutions.
For insights into the latest industry trends, innovations, and best practices, explore the Complete Water Solutions Blog, where Nathan shares valuable information on water filtration systems, sustainability, and water solutions engineering.
Give Us A Call (855) 787-4200 or Email info@complete-water.com












