The History of Reverse Osmosis
The History of Reverse Osmosis Technology
Osmosis has taken place for millions of years and happens in our bodies every day. But the history of reverse osmosis shows how this natural process was transformed into one of the most important purification technologies used today.
From early scientific discovery to modern industrial applications, reverse osmosis has evolved into a reliable solution for producing high-quality water across a wide range of industries. Understanding this history helps explain why reverse osmosis systems are trusted for critical operations where water quality cannot be compromised.
If your facility depends on consistent high-purity water, working with an experienced provider ensures you get a system designed for long-term performance – not just short-term results.
What is Reverse Osmosis?
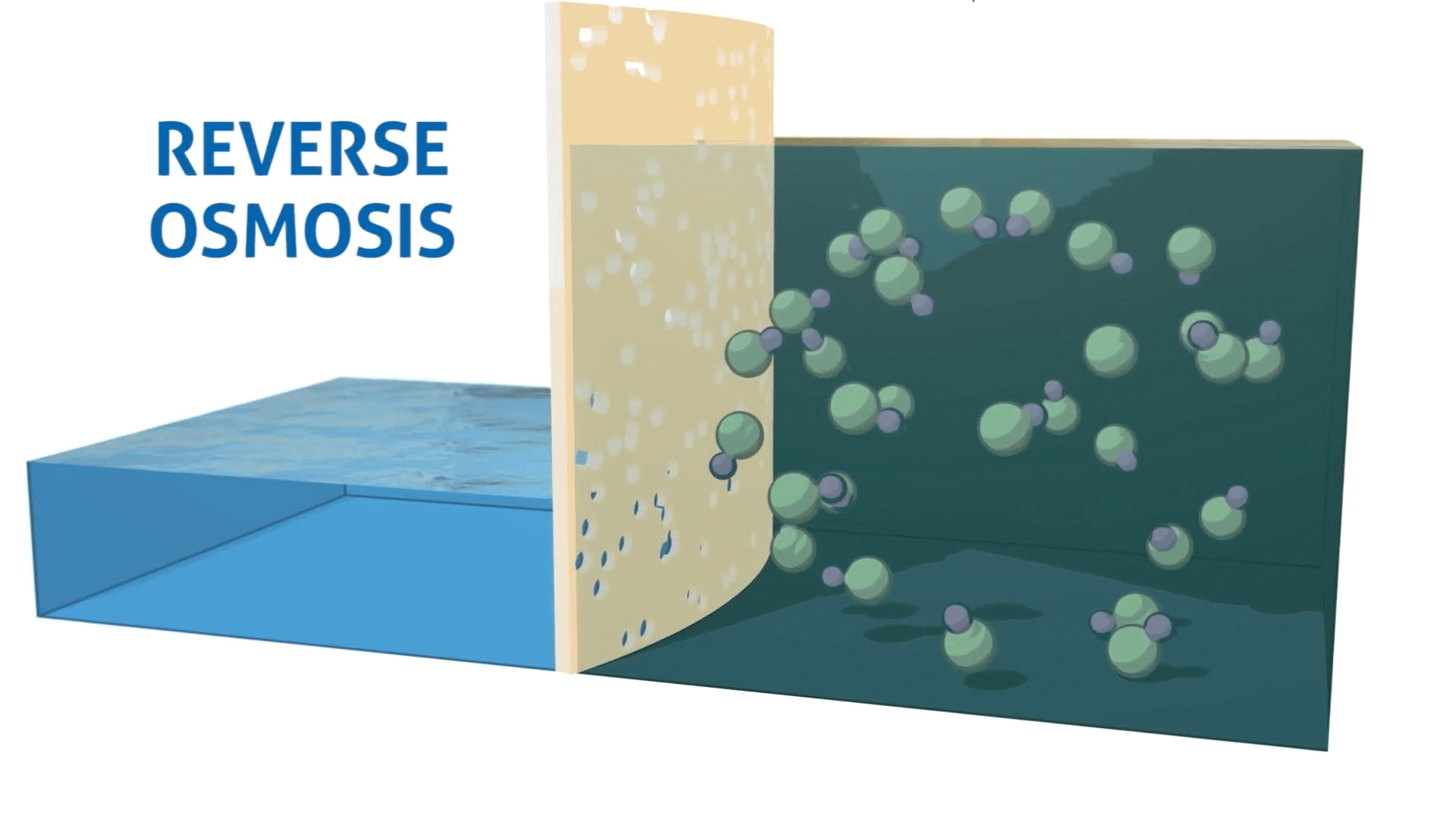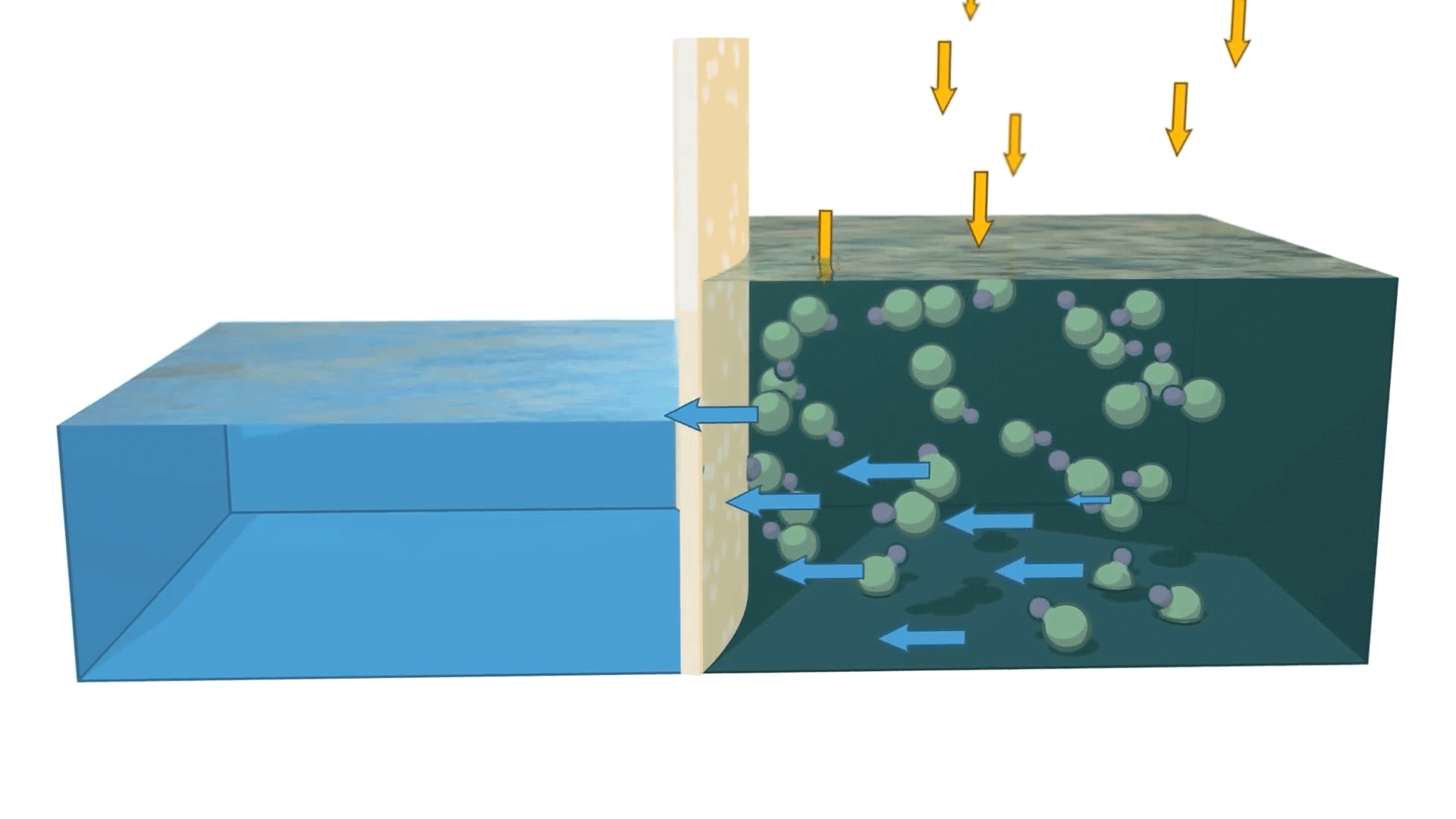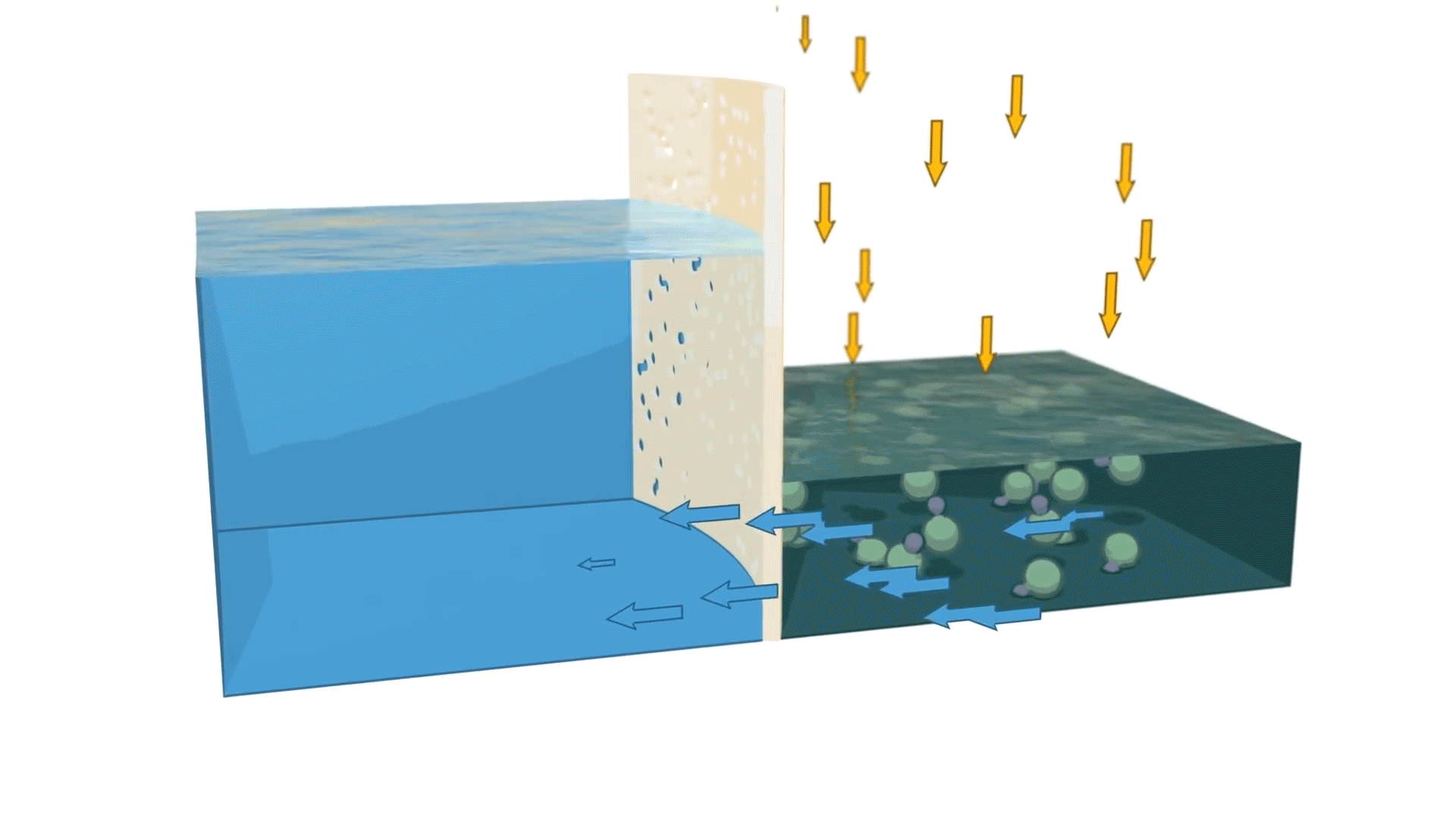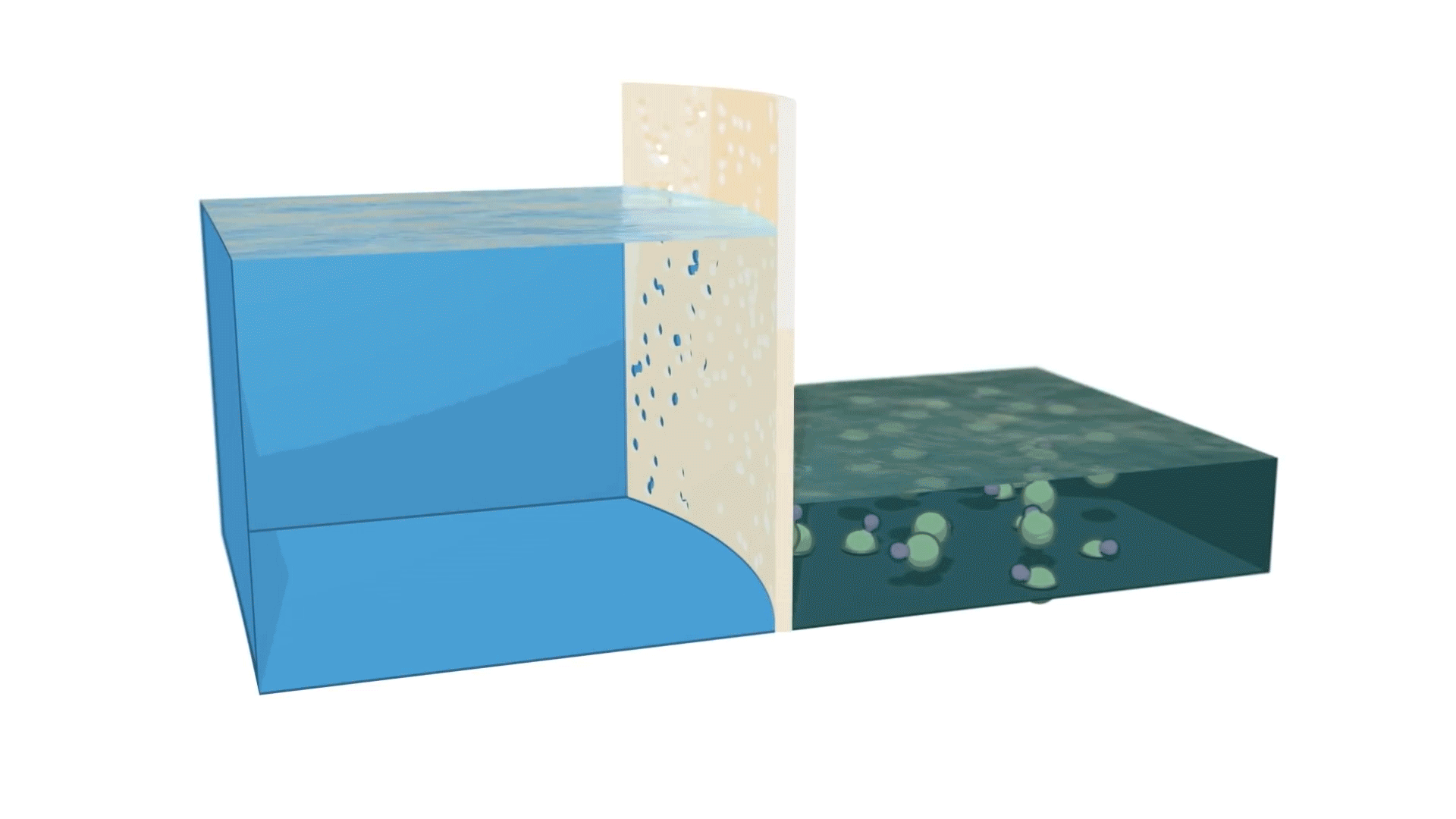
Reverse osmosis is the process of forcing a solvent from a region of high solute concentration through a semipermeable membrane into a region of low solute concentration by applying pressure in excess of the osmotic pressure.
In simpler terms, reverse osmosis removes dissolved solids and impurities by pushing water through a specialized membrane. The result is cleaner, more purified water that meets the demands of industrial, commercial, and municipal applications.
This process is especially important in environments where even small contaminants can lead to inefficiencies, product issues, or equipment damage.
When was Osmosis Discovered?
The process of osmosis through semipermeable membranes was first observed in a laboratory setting in 1748 by Jean-Antoine Nollet, using a pig’s bladder as a membrane. He proved that a solvent could pass selectively through a semipermeable membrane, demonstrating the natural osmotic pressure that occurs when equilibrium is reached.
While this discovery laid the foundation, it would take nearly two centuries before scientists could apply this concept in a way that would eventually lead to the history of reverse osmosis as we know it today.
Reverse Osmosis Through the Years
Over the next 200 years, osmosis remained largely a scientific observation until advancements in membrane technology made practical applications possible.
In 1949, the University of California Los Angeles began researching ways to use semipermeable membranes to desalinate seawater. Researchers from both UCLA and the University of Florida successfully produced fresh water from saltwater in the mid-1950s. However, the process was not yet commercially viable due to low efficiency.
That changed when Sidney Loeb and Srinivasa Sourirajan developed more effective membrane structures, making reverse osmosis practical for real-world use. By improving flow rates and salt rejection, they helped bring reverse osmosis out of the lab and into industrial applications.
By 2011, more than 15,000 desalination plants were operating or planned worldwide, highlighting how far the history of reverse osmosis had progressed.
In 1977, Cape Coral, Florida became the first municipality in the United States to use reverse osmosis on a large scale, operating a plant with an initial capacity of 3 million gallons per day. By 1985, it had grown into the largest low-pressure reverse osmosis plant in the world, producing 15 million gallons per day.
How Reverse Osmosis Is Used Today
As reverse osmosis technology advanced, it became a critical component in a wide range of industries. Today, it is used not only for purification but also for improving efficiency, reducing costs, and protecting equipment.
In boiler feedwater systems, reverse osmosis helps remove suspended and dissolved solids that can cause scaling and corrosion. This improves steam quality, enhances system efficiency, and reduces maintenance costs over time. Learn more about how proper water treatment impacts system performance by exploring our water treatment solutions.
For microelectronics and pharmaceutical manufacturing, reverse osmosis plays a key role in producing ultrapure water. These industries require extremely low levels of contaminants, and reverse osmosis provides a reliable way to meet those strict standards.
In the food and beverage industry, high-quality water is essential for both production and final product consistency. Reverse osmosis helps remove impurities while maintaining the desired characteristics needed for safe and consistent processing.
Water reuse and recycling have also become increasingly important as industries look for sustainable solutions. Reverse osmosis allows facilities to treat and reuse water, reducing waste and lowering overall water consumption.
In desalination, reverse osmosis transforms seawater and brackish water into usable water sources, making it an essential technology in regions where freshwater is limited.
ULTRAPURE WATER (MICROELECTRONICS AND PHARMACEUTICAL)
For microelectronics and pharmaceutical manufacturing, potable water supplied by a local municipality is not nearly pure enough. Their product specifications require water free of bacteria and organics, with dissolved solids levels that are 10,000 times lower than those found in potable drinking water. The first step in reducing the suspended solids levels is reverse osmosis where 98% of the influent water salts can be removed before additional treatment with advanced ion exchange or continuous deionization systems produce the ultrahigh purity water they need.
WATER REUSE & RECYCLING
The assumption that an unlimited quantity of affordable raw water is available to industry is no longer valid. Manufacturers are looking for alternate water sources to assure operational reliability and reduce potable water consumption. Spent process streams and plant wastewater effluent are now considered as possible water supplies and reverse osmosis can be an important technology in reducing dissolved solids levels and enabling these water streams to be reused and recycled back into the plant.
BOILER FEEDWATER
Industries that rely on steam as a driving force for turbines, an energy source for heating, or product processing know that suspended and dissolved solids in a steam generator cause scale and can lead to tube failure. If you have poor quality boiler feedwater, it will increase energy consumption, reduce steam quality and purity, and can reduce both production rates and product quality. Including reverse osmosis systems in the boiler feedwater makeup circuit to replace or pretreat for ion exchange increases production reliability and significantly reduces operating and maintenance costs.
INGREDIENT WATER (FOOD & BEVERAGE)
High quality water is a critical ingredient in the food and beverage processing industry. Not only is it the obvious foundation of the bottled water industry but it is also important in beer production, where high quality water is used in the proofing process for alcohol beverage production. Meat and poultry processing as well as fresh pack and processed food production also need high quality water requirements. RO product water can meet all these needs. In addition to the ability to reduce dissolved solids, reverse osmosis can also remove pollutants and microorganisms.
DESALINATION
The earth is covered primarily by water, however, much of this water has a high salt content and cannot be used for either human consumption or industrial processing. However, in certain regions, the only available water source is either brackish water or seawater. Reverse osmosis systems can reduce the TDS level of brackish water and seawater to potable levels and further treatment can generate the higher purity waters needed for industry, medical care, and institutional use.
Why Reverse Osmosis Expertise Matters
The history of reverse osmosis shows that this technology has continuously evolved—and choosing the right system today requires more than basic knowledge.
Proper system design, installation, and maintenance all play a critical role in long-term performance. Without the right approach, systems can become inefficient, costly, and prone to failure.
At Complete Water Solutions, our team brings decades of experience in water treatment, helping businesses implement reverse osmosis systems that are built for reliability and efficiency. From system selection to ongoing service, we ensure your water quality supports your operation—not holds it back.
Get the Right Reverse Osmosis Solution for Your Facility
If your current system is struggling to keep up, or you’re planning for future growth, now is the time to take action. The right reverse osmosis system can improve efficiency, reduce costs, and protect your equipment for years to come.
Contact our team today to schedule a consultation or service call and discover how modern reverse osmosis solutions can support your operation.